Baden et al.1 report on a phase 3 clinical trial of the mRNA-1273 vaccine against SARS-CoV-2, and they provide information on immediate injection-site reactions, which were observed in 84.2% of the participants after the first dose. The trial also showed that delayed injection-site reactions (defined in that trial as those with an onset on or after day 8) occurred in 244 of the 30,420 participants (0.8%) after the first dose and in 68 participants (0.2%) after the second dose. These reactions included erythema, induration, and tenderness. The reactions typically resolved over the following 4 to 5 days. However, these reactions were not further characterized, and links between reactions after the first dose and those after the second dose were not provided to inform clinical care.
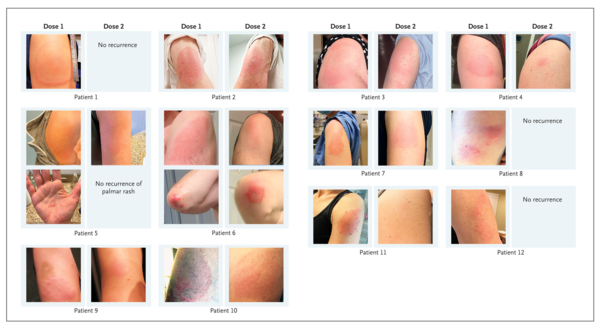
We have also observed delayed large local reactions to the mRNA-1273 vaccine, with a median onset on day 8 (range, 4 to 11) after the first dose. These reactions had a variable appearance (Figure 1). Here, we report on a series of 12 patients with these reactions, all of which appeared near the injection site after complete resolution of the initial local and systemic symptoms associated with vaccination. Five of the reactions were grade 3 plaques (≥10 cm in diameter) (Table 1). Some patients had concurrent systemic adverse effects, and among these patients, 2 had additional skin findings. Most patients received treatment for their symptoms (e.g., with ice and antihistamines). Some patients received glucocorticoids (topical, oral, or both), and 1 patient received antibiotic therapy for presumptive cellulitis. The symptoms resolved a median of 6 days after onset (range, 2 to 11).
Our suspicion of delayed-type or T-cell-mediated hypersensitivity was supported by skin-biopsy specimens obtained from a patient with a delayed large local reaction who was not among the 12 patients described here. Those specimens showed superficial perivascular and perifollicular lymphocytic infiltrates with rare eosinophils and scattered mast cells (see Fig. S1 in the Supplementary Appendix, available with the full text of this letter at NEJM.org).
Given that neither local injection-site reactions nor delayed-type hypersensitivity reactions are contraindications to subsequent vaccination,2 all 12 patients were encouraged to receive the second dose and completed their mRNA-1273 vaccination course. Although half the patients did not have a recurrence of large local reactions, three patients had recurrent reactions that were similar to those after the initial dose, and three patients had recurrent reactions that were of a lower grade than those after the initial dose. The median onset of cutaneous symptoms after the second dose (day 2; range, 1 to 3) was earlier than that after the first dose (Table 1).
Clinicians may not be prepared to address delayed local reactions to the mRNA-1273 vaccine. Given the scale-up of mass vaccination campaigns across the world, these reactions are likely to generate concerns among patients and requests for evaluation. These reactions have not been consistently recognized, guidance regarding the second dose of vaccine has varied, and many patients have unnecessarily received antibiotic agents. We hope this letter encourages additional reporting and communication regarding the epidemiologic characteristics, causes, and implications of these delayed cutaneous reactions, since this information might allay the concerns of patients, encourage completion of vaccination, and minimize the unnecessary use of antibiotic agents.
Badenらは SARS-CoV-2 に対する mRNA-1273 ワクチンの第 3 相臨床試験を報告し、彼らは最初の投与後の参加者の 84.2% で観察された即時の注射部位反応についての情報を提供している。また、この試験では、遅延型の注射部位反応(8 日目以降に発症したものとしてこの試験で定義)が 30,420 人の参加者のうち 244 人(0.8%)で 1 回目の投与後に、68 人(0.2%)で 2 回目の投与後に発生したことが示されている。これらの反応には、紅斑、硬結、圧痛が含まれていた。これらの反応は通常、その後4~5日で消失した。しかし、これらの反応はさらに特徴づけられておらず、1回目の投与後の反応と2回目の投与後の反応の間の関連性は、臨床ケアへの情報提供のために提供されなかった。
我々はまた、mRNA-1273ワクチンに対する遅発性の大規模な局所反応も観察しており、中央値は初回投与後8日目(範囲は4~11日目)の発症であった。これらの反応は変化に富んだものであった(図1)。ここでは、これらの反応を呈した12人の患者のシリーズについて報告する。これらの反応はすべて、ワクチン接種に関連した初期の局所および全身症状が完全に消失した後、注射部位付近に出現した。これらの反応のうち5人はグレード3のプラーク(直径10cm以上)であった(表1)。何人かの患者は全身性の副作用を併発しており、これらの患者のうち2人は追加の皮膚所見を有していた。ほとんどの患者は症状に対する治療を受けていた(例:氷や抗ヒスタミン薬)。一部の患者はグルココルチコイド(外用薬、経口薬、またはその両方)を投与され、1人の患者は蜂巣炎を想定され、抗生物質療法を受けた。症状は発症から中央値で6日後に消失した(範囲は2~11日)。
遅延型またはT細胞を介した過敏症に対する疑いは、ここに記載された12人の患者の中には含まれない、遅延型の大規模局所反応を起こした患者から得られた皮膚バイオプシー標本によって裏付けられた。これらの検体は、まれな好酸球と散在したマスト細胞を伴う表在性の血管周囲および毛包周囲リンパ球浸潤を示した(NEJM.orgで本レターの全文と一緒に入手可能な補足資料の図S1を参照のこと)。
局所的な注射部位反応も遅延型過敏症反応も、その後のワクチン接種の禁忌ではないことから、 12 人の患者全員に 2 回目の接種を勧め、mRNA-1273 ワクチン接種コースを終了した。半数の患者では大規模な局所反応の再発はなかったが,3名の患者で初回接種後と同程度の再発があり,3名の患者では初回接種後よりも悪性度の低い再発があった.2回目の投与後(2日目;範囲、1~3)の皮膚症状の発現の中央値は、1回目の投与後よりも早かった(表1)。
臨床医は、mRNA-1273ワクチンに対する遅延した局所反応に対応する準備ができていない可能性がある。世界中で行われている大量接種運動の規模拡大を考えると、これらの反応は患者の間に懸念を生じさせ、評価を求めることになるだろう。これらの反応は一貫して認識されておらず、2回目のワクチン接種に関するガイダンスも様々であり、多くの患者が不必要に抗生物質を投与されている。私たちは、この文書が、これらの遅延した皮膚反応の疫学的特徴、原因、および意味合いに関する追加の報告とコミュニケーションを奨励するものであることを願っている。
Figure 1. Delayed Cutaneous Reactions to mRNA-1273 Vaccine.
https://www.nejm.org/doi/full/10.1056/NEJMc2102131