On December 20, 2020, Israel initiated a national vaccination program against COVID-19. One prioritized group was health care workers, many of whom are breastfeeding women. Despite the fact that the vaccine trial did not include this population and no other vaccine-related safety data had been published, breastfeeding women belonging to risk groups were encouraged to receive the vaccine. The Centers for Disease Control and Prevention has also recommended that breastfeeding women belonging to vaccine-target groups be immunized. We investigated whether maternal immunization results in secretion of SARS-CoV-2 antibodies into breast milk and evaluated any potential adverse events among women and their infants.
We conducted a prospective cohort study of a convenience sample of breastfeeding women (either exclusive or partial) belonging to vaccine-target groups who chose to be vaccinated. Participants were recruited from all of Israel between December 23, 2020, and January 15, 2021, through advertisements and social media. All participants received 2 doses of the Pfizer-BioNTech vaccine 21 days apart. Breast milk samples were collected before administration of the vaccine and then once weekly for 6 weeks starting at week 2 after the first dose. Samples were kept frozen pending analysis. IgG levels were detected by the Elecsys Anti-SARS-CoV-2 S serology assay and read on the Cobas e801 analyzer with a level of more than 0.8 U/mL considered positive (La Roche Ltd) and IgA with the EUROIMMUN AG Anti-SARS-CoV-2 S Kit with an extinction ratio of samples over calibrator of more than 0.8 considered positive (Supplement). At enrollment, maternal and infant demographic information was collected, followed by weekly questionnaires coupled to breast milk collection soliciting information about interim well-being and vaccine-related adverse events. The study was approved by the Shamir Medical Center Institutional Review Board; written informed consent was obtained from mothers.
Changes in the proportion of participants with positive test results and in antibody levels during the study were evaluated using paired-sample t tests, comparing antibody levels at each point with the baseline and correcting for multiple testing using the Benjamini-Hochberg procedure. A 2-sided significance threshold was set at P < .05. Analyses were performed with R version 3.6
Eighty-four women completed the study, providing 504 breast milk samples. Women were a mean (SD) age of 34 (4) years and infants 10.32 (7.3) months (Table).
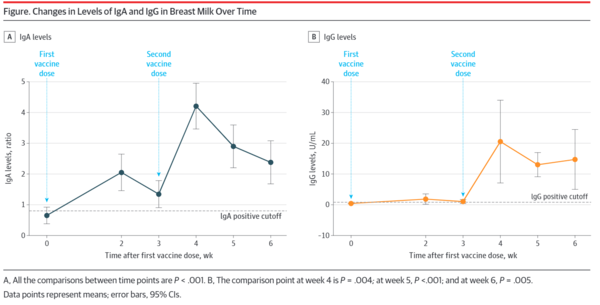
Mean levels of anti-SARS-CoV-2-specific IgA antibodies in the breast milk increased rapidly and were significantly elevated at 2 weeks after the first vaccine (2.05 ratio; P < .001), when 61.8% of samples tested positive, increasing to 86.1% at week 4 (1 week after the second vaccine). Mean levels remained elevated for the duration of follow-up, and at week six, 65.7% of samples tested positive. Anti-SARS-CoV-2-specific IgG antibodies remained low for the first 3 weeks, with an increase at week 4 (20.5 U/mL; P = .004), when 91.7% of samples tested positive, increasing to 97% at weeks 5 and 6 (Figure).
No mother or infant experienced any serious adverse event during the study period. Forty-seven women (55.9%) reported a vaccine-related adverse event after the first vaccine dose and 52 (61.9%) after the second vaccine dose, with local pain being the most common complaint (Table). Four infants developed fever during the study period 7, 12, 15, and 20 days after maternal vaccination. All had symptoms of upper respiratory tract infection including cough and congestion, which resolved without treatment except for 1 infant who was admitted for neonatal fever evaluation due to his age and was treated with antibiotics pending culture results. No other adverse events were reported.
This study found robust secretion of SARS-CoV-2 specific IgA and IgG antibodies in breast milk for 6 weeks after vaccination. IgA secretion was evident as early as 2 weeks after vaccination followed by a spike in IgG after 4 weeks (a week after the second vaccine). A few other studies have shown similar findings in women infected with COVID-19. Antibodies found in breast milk of these women showed strong neutralizing effects, suggesting a potential protective effect against infection in the infant.
The study has limitations. First, no functional assays were performed. However, previous studies have showed neutralizing capacities of the same antibodies as measured for this study. Second, serum antibody testing or SARS-CoV-2 real-time reverse-transcriptase polymerase chain reaction testing were not performed, which would have provided interesting correlates.
2020年12月20日、イスラエルはCOVID-19に対する全国的なワクチン接種プログラムを開始した。優先されたグループの1つは医療従事者で、その多くは母乳育児中の女性であった。ワクチン試験にはこの集団が含まれておらず、その他のワクチン関連の安全性データも発表されていなかったにもかかわらず、リスクグループに属する授乳中の女性にワクチンの接種が勧められた。また,米国疾病対策予防センターは,ワクチン対象グループに属する母乳育児中の女性にも接種を推奨している。我々は,母親の予防接種によってSARS-CoV-2抗体が母乳中に分泌されるかどうかを調べ,女性とその乳児に起こりうる有害事象を評価した。
ワクチン接種対象グループに属する母乳育児中の女性(完全母乳または部分母乳)のうち,ワクチン接種を選択した便宜的なサンプルを対象とした前向きコホート研究を行った。参加者は、2020年12月23日から2021年1月15日の間に、広告やソーシャルメディアを通じてイスラエル全土から募集した。すべての参加者は、ファイザー・バイオンテック社製のワクチンを21日間隔で2回接種した。母乳サンプルは、ワクチン投与前に採取し、その後、初回投与後2週目から6週間にわたり週1回採取した。サンプルは分析するまで冷凍保存した。IgGレベルはElecsys Anti-SARS-CoV-2 S serology assayで検出し、Cobas e801アナライザーで読み取り、0.8 U/mL以上を陽性とした(La Roche Ltd)。IgAはEUROIMMUN AG Anti-SARS-CoV-2 S Kitで検出し、キャリブレーターに対するサンプルの extinction ratioが0.8以上を陽性とした(Supplement)。登録時に母親と乳児の人口統計学的情報を収集した後、母乳を採取する際に週1回のアンケートを実施し、中間的な健康状態やワクチン関連の有害事象に関する情報を収集した。この研究はShamir Medical Center Institutional Review Boardの承認を得ており、母親からは書面によるインフォームドコンセントを得た。
試験期間中の検査結果が陽性であった参加者の割合と抗体レベルの変化は、ペアサンプルt検定を用いて評価し、各時点の抗体レベルをベースラインと比較し、Benjamini-Hochberg法を用いて多重検定を補正した。2辺の有意差の閾値はP < 0.05とした。解析にはRバージョン3.6を使用した。
84名の女性が調査に参加し、504個の母乳サンプルを提供した。女性の平均(SD)年齢は34(4)歳、乳児は10.32(7.3)カ月であった(表)。
母乳中の抗SARS-CoV-2特異的IgA抗体の平均値は急速に上昇し,1回目のワクチン接種後2週間で61.8%が陽性となり(2.05比,P < 0.001),4週目(2回目のワクチン接種後1週間)には86.1%にまで上昇した。平均値はフォローアップ期間中も上昇し続け、6週目には65.7%の検体が陽性となった。抗SARS-CoV-2特異的IgG抗体は,最初の3週間は低値であったが,4週目に上昇し(20.5 U/mL,P = 0.004),91.7%の検体が陽性となり,5週目と6週目には97%まで上昇した(図)。
試験期間中,重篤な有害事象を経験した母親と乳児はいなかった。ワクチン関連の有害事象を報告したのは、1回目のワクチン接種後に47名(55.9%)、2回目のワクチン接種後に52名(61.9%)で、局所的な痛みが最も多く報告された(表)。母体のワクチン接種後7、12、15、20日目の調査期間中に、4名の乳児が発熱した。全員が咳や鼻づまりなどの上気道感染症の症状を呈していたが,年齢的に新生児発熱評価のために入院し,培養結果を待って抗生物質で治療した1名の乳児を除いて,治療なしで回復した。その他の有害事象は報告されなかった。
本研究では、SARS-CoV-2に特異的なIgAおよびIgG抗体が、ワクチン接種後6週間にわたって母乳中にしっかりと分泌された。IgAの分泌はワクチン接種後2週間で明らかになり、続いて4週間後(2回目のワクチン接種後1週間)にIgGのスパイクが見られた。他のいくつかの研究では、COVID-19に感染した女性で同様の所見が得られている。これらの女性の母乳から検出された抗体は強い中和作用を示し、乳児の感染に対する保護効果の可能性を示唆している。
この研究には限界がある。まず、機能的アッセイが行われていない。しかし、過去の研究では、本研究で測定したのと同じ抗体の中和能力が示されている。第二に、血清抗体検査やSARS-CoV-2リアルタイム逆転写酵素ポリメラーゼ連鎖反応検査を実施していれば、興味深い相関関係が得られたと思われる。
https://jamanetwork.com/journals/jama/fullarticle/2778766